Containment in ADC Facilities
Containment Evaluation and Design Considerations
Produktion
Abstract
The present article deals with the process design of a high containment pharmaceutical production facility. It depicts how to evaluate the containment performance of this kind of production, discussing the features of the systems at different levels of details: device level, equipment level, room level and organization level. For each level, some considerations and recommendations are provided. Subsequently, the algorithm to execute the calculation is provided and commented.
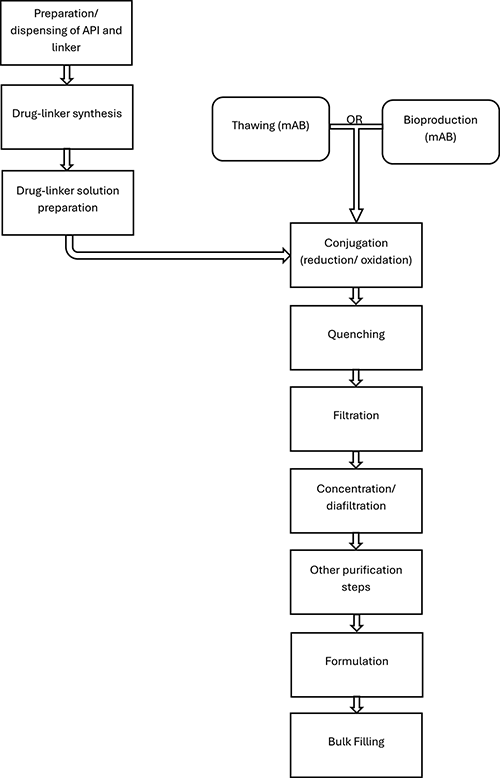
The 2nd part of the article illustrates containment topics related to Antibody Drug Conjugate (ADC) production. Finally, the author describes a possible design for an ADC production facility, which optimizes the containment performance of the system and gives additional substantial advantages compared to traditional solutions. An example of such design is provided.
Correspondence:
Pietro Virgilio
Howorth Air Technology Limited, Lomax Way, Bolton, BL5 1FQ, UK
Pietro.Virgilio@howorthgroup.com
 | Ing. Pietro Virgilio graduated in Chemical Engineering at the Politecnico di Milano (Italy) in 1996 and has 28 years’ experience in the design of pharma facilities. His main focuses are isolator technology, containment, decontamination, sterilization and pure fluids. He worked in the process department of different engineering companies on large biopharma projects in Europe for many years. He is a ISPE member and has published papers with a few Italian pharma associations (ASCCA, AFI). He |
Schließen Sie hier ein Abonnement ab und profitieren Sie von den vielseitigen Nutzungsmöglichkeiten.