Header
Ihr Suchergebnis
Sie recherchieren derzeit unangemeldet.Melden Sie sich an (Login) um den vollen Funktionsumfang der Datenbank nutzen zu können.
Sie suchen in allen Bereichen nach dem Autor Futschik M.
In der Rubrik Zeitschriften haben wir 3 Beiträge für Sie gefunden
-
Comparison of Electronic Batch Recording Systems in the pharmaceutical industry
Rubrik: IT
(Treffer aus TechnoPharm, Nr. 01, Seite 44 (2017))
Comparison of Electronic Batch Recording Systems in the pharmaceutical industry / Part 2 * Part 1 of this article was published in TechnoPharm 6, Nr. 6, 352–359. · Futschik M · Roche Diagnostics GmbH, Mannheim
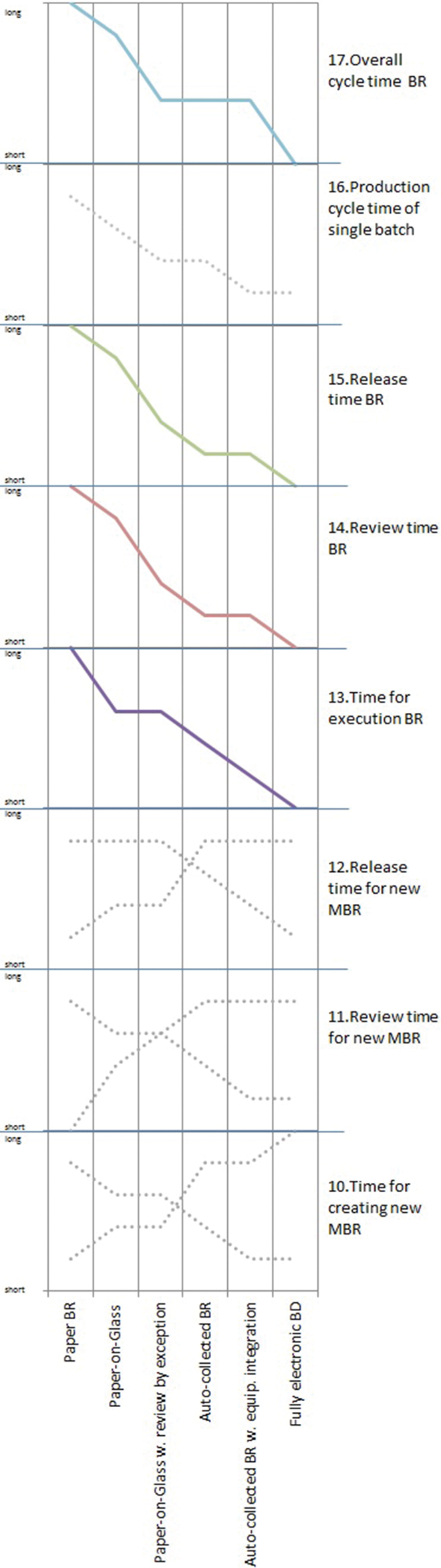
The study shows a divergent picture in regards to process time criteria as illustrated in Fig. 8 . While some criteria follow a clear trending others are disordered and show huge variation. The non-general criteria are interesting since they tend to oppose against general benefits often promoted by software providers. The time required for creating, reviewing and releasing MBR templates for a new product was evaluated differently by participants. During the study it became clear that these criteria are driven by intra-organizational aspects of the particular company such as the number and complexity of existing processes, the number of production lines ...
-
Comparison of Electronic Batch Recording Systems in the pharmaceutical industry
Rubrik: IT
(Treffer aus TechnoPharm, Nr. 06, Seite 352 (2016))
Comparison of Electronic Batch Recording Systems in the pharmaceutical industry / Part 1 · Futschik M · Roche Diagnostics GmbH, Mannheim
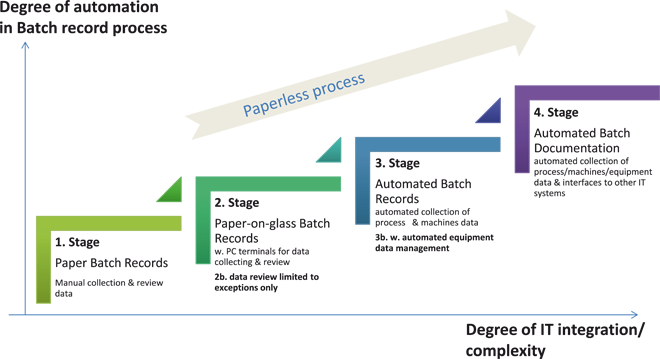
Electronic batch recording solution paperless manufacturing MES In pharmaceutical operations the Batch Record (BR) is the centerpiece of the manufacturing documentation. From a supply chain perspective, the document is as important as the product. Without complete and approved batch documentation no distribution or sale of the product is allowed. From an operations perspective, it proofs the proper handling of each step of the production including operators, machines, materials and processes. The batch record documentation process can be understood as a regulatory required end-to-end process integrating the creation of templates, the collection and recording of data throughout the production of a ...
-
Evaluation of Electronic Batch Recording Systems
Rubrik: Fachthemen
(Treffer aus pharmind, Nr. 04, Seite 494 (2016))
Evaluation of Electronic Batch Recording Systems / Organizational aspects · Futschik M · Roche Pharmaceuticals, Mannheim
Previous literature about paperless documentation in the pharmaceutical industry showed that the implementation of Electronic Batch Recording Systems (EBRS) does not provide an optimization per se [ 1 – 4 ]. The benefits of EBRS deployment in operations are in conflict with massive change management efforts and investment costs required for system deployment. This means that a careful and systematic evaluation is necessary to justify the implementation. Unfortunately, existing evaluations’ examples were found to be incomplete since they primarily rely on using investment costs and technical feasibility, but do not consider the organizational characteristics of organizations which determine the change ...