Header
Ihr Suchergebnis
Sie recherchieren derzeit unangemeldet.Melden Sie sich an (Login) um den vollen Funktionsumfang der Datenbank nutzen zu können.
Sie suchen in allen Bereichen nach dem Autor Groß-Weege C.
In der Rubrik Zeitschriften haben wir 2 Beiträge für Sie gefunden
-
Rapid Control Prototyping for Continuous Pharmaceutical Tablet Production Processes
Rubrik: Technik
(Treffer aus pharmind, Nr. 02, Seite 290 (2017))
Groß-Weege C | Rehbaum H | Abel D
Rapid Control Prototyping for Continuous Pharmaceutical Tablet Production Processes / Groß-Weege et al. • Rapid Control Prototyping · Groß-Weege C, Rehbaum H, Abel D · 1Institut für Regelungstechnik, RWTH Aachen University, Aachen (Germany) und 2L.B. Bohle Maschinen + Verfahren GmbH, Ennigerloh (Germany)
More recently, continuous processes and the idea of continuous production systems have received increased interest in the pharmaceutical industry for solid dosage forms. Batch-oriented production processes – currently state of the art in the pharmaceutical industry – are known to have limited flexibility, higher risks of waste production and reduced capabilities of in-process monitoring. Additionally, regulatory facilities stimulated the pharmaceutical industry by several approaches, including Quality by Design (QbD) and Process Analytical Technology (PAT), to raise its interest in an in-depth understanding of process and product characteristics. To overcome these limitations and implement new quality approaches, integrated continuous production systems ...
-
Automatisierung kontinuierlicher Herstellungsprozesse
Rubrik: Automation
(Treffer aus TechnoPharm, Nr. 06, Seite 336 (2016))
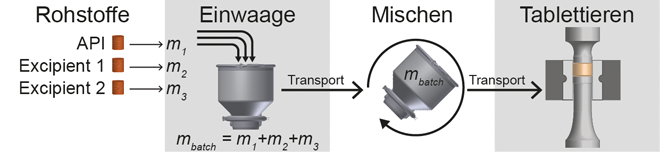
Automatisierung kontinuierlicher Herstellungsprozesse / Herausforderung bei der kontinuierlichen Produktion pharmazeutischer Feststoffe · Rehbaum H, Groß-Weege C · L.B. Bohle Maschinen + Verfahren GmbH, Ennigerloh und Institut für Regelungstechnik an der RWTH Aachen, Aachen
Continuous Manufacturing Kontinuierliche Direktverpressung Automatisierung QbD OSD Secondary Manufacturing Im Zuge der Initiativen zur Qualitätssteigerung und vor allem getrieben durch die US-amerikanische Regulierungsbehörde FDA (U.S. Food and Drug Administration) rückt die kontinuierliche Fertigung als nächste Generation der Herstellung fester Arzneiformen in den Vordergrund. Unter kontinuierlicher Fertigung wird dabei die vollintegrierte Verarbeitung der Rohstoffe – d. h. Wirkstoffe (Active Pharmaceutical Ingredient – API) und Excipients – zu Tabletten als verkaufsfähige Produkte verstanden. Sowohl Pharmaunternehmen als auch Maschinenhersteller haben diesen Trend aufgegriffen und arbeiten an technischen Lösungen zur Erreichung dieses Ziels. Neben der Entwicklung neuartiger Anlagen für die kontinuierliche Produktion steht nun insbesondere ...